台灣尖端先進生技醫藥股份有限公司TAIWAN ADVANCE BIO-PHARMACEUTICAL INC.
Hematopoietic stem cell research and development technology has won professional recognition at home and abroad
With the advancement of medical technology, stem cell applications are becoming lighter. The cutting-edge doctors who focus on research and development also strive to break through the restrictions and develop stem cell proliferation technology. Today, excellent research and development results are not only recognized by international journals, but also pre-clinical trials have been successfully completed. In 2012, it won the national new award. The stem cell proliferation technology of cutting-edge doctors is to promote the in vitro proliferation of stem cells through TAT-HOXB4 hematopoietic stem cell growth factor, which is 8 times in 6 days. Technical patents are spread throughout Europe, America and Asia.
Testing service
Food safety testing
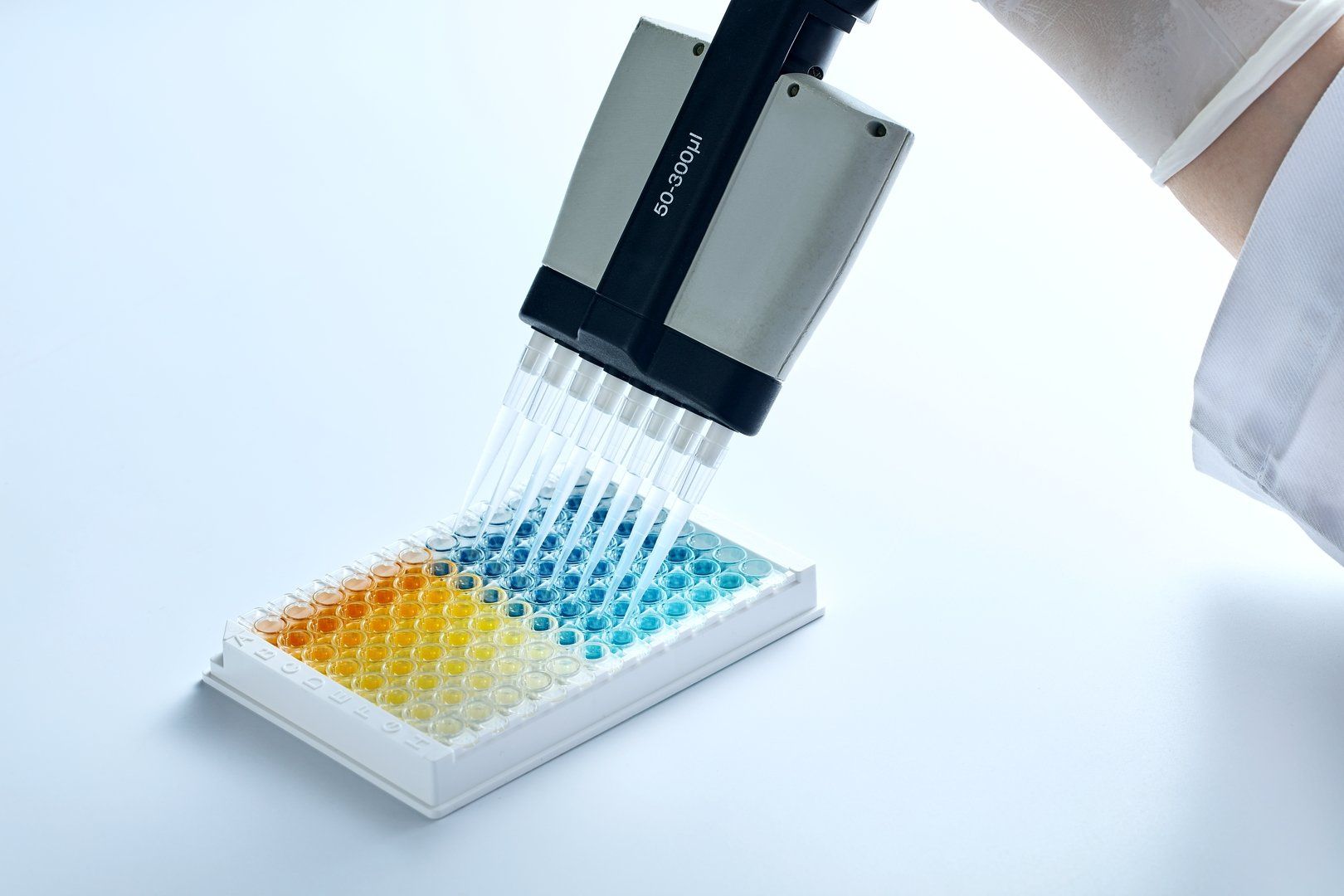
The cutting-edge doctors have a food certification laboratory, which mainly uses ELISA and Rapid test. The current test body includes meat, raw milk, internal organs, eggs, honey, and feed. Provide accurate, fast, and cost-effective food testing solutions.
Drug and drug abuse testing
The drug abuse testing service is a check for drug abuse. The laboratory of the company was examined and approved by the Weifu Department of the Executive Yuan in accordance with the “Administration and Management Points for Substance Abuse Drugs”. The legal appraisers of the Procuratorate of the High Court have also accepted the inspection and analysis of the drug components in the urine.
More information
New drug development and cell preparation
The cutting-edge medical research and development team is dedicated to the development of hematopoietic stem cell proliferation technology, and established the [Hb4Cord Hematopoietic Stem Cell Proliferation Technology Platform]. This platform has several innovative features: high-efficiency frozen umbilical cord blood thawing purification recovery rate, high magnification to increase the number of hematopoietic stem cells, A high proportion of effective hematopoietic stem cell (CD34 CD38-cell) population, high-recovery freeze-thaw preservation technology, non-viral or gene introduction systems are safer.
latest news

隨著國內食品安全意識升溫與檢測法規趨嚴,食品檢測與快速篩檢市場需求持續擴大,尖端醫(4186)近年積極深耕食安健康與毒物分析領域,在毒品證物檢驗業務帶動下,114年度營收與獲利表現同步成長,並進一步布局馬鈴薯「龍葵鹼(Solanine)」檢測與快篩技術,搶攻新興食安檢測商機。 在毒物檢測業務方面,因應警政署積極強化毒駕取締趨勢,尖端醫 114 年毒品證物檢驗收件數較 113 年顯著成長 60%,帶動相關檢測業務營收提升 15%,展現在毒品檢驗與司法鑑定市場的領導地位。 受各項核心業務成長挹注,尖端醫 114 年度整體獲利達 2,804.7萬元,每股稅後盈餘(EPS)0.31 元,高於 113 年的 2,287.5萬元與 EPS 0.25 元,顯示檢測事業已成為公司成長動能之一。 近期國內因馬鈴薯龍葵鹼與總配醣生物鹼議題引發市場高度關注,由於相關天然毒素若攝取過量,恐對人體健康造成影響,因此食品業者對於精準檢測與即時篩檢工具需求快速升高,然而,目前國內具備相關檢測量能與認證能力的機構仍相對有限,市場存在明顯技術缺口,因應市場需求,尖端醫旗下食安健康事業處將投入馬鈴薯總配醣生物鹼檢測技術開發,並同步進行標準檢測方法建置與驗證評估,希望未來能提供食品加工、餐飲與供應鏈業者更完整的食安風險管理方案。 尖端醫指出,公司長期建置完整精密分析平台,包括氣相層析質譜(GC-MS)與液相層析串聯質譜(LC-MS/MS)等設備,具備導入衛福部公告檢測方法能力,未來將針對馬鈴薯中的主要毒素α-solanine與α-chaconine進行高精準定量分析,以提升檢測準確度與可靠性,除了精密儀器分析外,公司亦同步推動快速篩檢技術開發,整合酵素免疫分析(ELISA)與免疫層析(Lateral Flow)技術,評估開發龍葵鹼快篩產品。若後續技術驗證成熟,未來可望導入食品產線、物流通路及第一線品管流程,協助業者快速辨識風險產品,縮短檢測時間並提升管理效率。 尖端醫表示,食安健康事業處目前已建立從抗體開發、試劑設計、製程優化到量產導入的完整整合能力,可快速回應新興食品安全議題並推動技術商品化,公司將持續以「精密檢驗+快速篩檢」雙軌策略,深化食品安全檢測服務與產品布局,在全球食安法規日益嚴格與消費者健康意識提升趨勢下,高準確度、高效率且具即時性的檢測方案,將成為食品產業重要基礎工具,也有助帶動台灣檢測產業與生技應用市場持續成長。 以上新聞,分享於 ETtoday財經雲 ,內容及版權歸該媒體所有。

為強化毒品防制與道路安全管理,內政部警政署自11月20日起正式推動「毒品唾液快篩執法」,全國警察機關同步全面上路。此項新制被視為我國毒駕查緝的重要制度升級,不僅大幅提升第一線員警的即時判定能力,也同步帶動後端毒品檢驗與司法鑑定需求快速攀升。市場普遍預期,在政策推動與查緝量增加的雙重效應下,相關檢測產業將迎來新一波成長動能,其中長期深耕毒品檢測領域的尖端醫(4186)可望成為主要受益者之一。 過去毒駕取締主要仰賴尿液或血液檢測,不僅採檢程序較為繁瑣,且在時間與現場判定上存在一定限制。隨著唾液快篩技術納入執法流程,員警可於現場快速進行初步檢測,顯著提升執法效率與即時性,並降低漏判風險。尤其針對近年快速擴散的新興毒品,如依托咪酯(俗稱「喪屍菸彈」),因其代謝速度快、過往難以透過傳統檢測方式掌握,如今透過唾液檢測能有效提高查緝成功率,對整體毒品防制工作具有關鍵意義,在檢測體系方面,目前於北區取得衛生福利部食品藥物管理署認可、可執行毒品尿液確認檢驗的民間機構中,僅尖端醫具備完整受理資格。隨著新制上路後查緝件數預期增加,後端送驗需求將同步放大,使具備實驗室檢測能力的業者直接受惠,尖端醫憑藉其既有技術基礎與法規資格,將在此波需求擴張中占據有利位置。 快篩產品放量 成為營收新引擎 其次,在企業市場(B2B)方面,政策強化將促使高風險產業加速導入員工藥物濫用管理制度。包括運輸業、物流業及製造業等,基於安全與法規要求,將提高對員工定期檢測的需求。此一趨勢有助於擴大毒品檢測服務在企業端的應用場景,為尖端醫開啟新的市場成長空間。第三,在產品銷售方面,隨著唾液快篩具備明確法源依據,警政機關與教育單位將更積極採用相關檢測工具,帶動唾液快篩試劑需求快速成長。